How I Can Buy Oxycontin 10mg Pills Online
Buy oxycontin 10mg pills online in the states of the USA and Canada with overnight shipping. We are providing you overnight shipping service. We will will delivery your oxycontin 10mg medicine at your doorsteps without any Rx required. We are selling best quality of oxycontin 10mg pills at a cheap price. We are accept all kinds of online payment like net banking, PayPal, credit card and debit card and cash on delivery option is available here.
What Is Oxycontin
OxyContin Neo (oxycodone hydrochloride controlled-release) is an opioid analgesic supplied in 10 mg, 20 mg, 40 mg, 60 mg and 80 mg tablets for oral administration. The tablet strengths describe the amount of oxycodone per tablet as the hydrochloride salt.
The structural formula for oxycodone hydrochloride is: C18H21NO4·HCl. MW: 351.83. The chemical name is 4, 5α-epoxy-14-hydroxy-3-methoxy-17-methylmorphinan-6-one hydrochloride. Oxycodone is a white, odorless crystalline powder derived from the opium alkaloid, thebaine. Oxycodone hydrochloride dissolves in water (1 g in 6 to 7 mL). It is slightly soluble in alcohol (octanol water partition coefficient 0.7). OxyContin Neo 10 mg (oxycodone hydrochloride): Each contains oxycodone hydrochloride 10 mg equivalent to 9 mg oxycodone base. OxyContin Neo 20 mg (oxycodone hydrochloride): Each contains oxycodone hydrochloride 20 mg equivalent to 18 mg oxycodone base. OxyContin Neo 40 mg (oxycodone hydrochloride): Each contains oxycodone hydrochloride 40 mg equivalent to 36 mg oxycodone base. Excipients/Inactive Ingredients: Butylated hydroxytoluene (BHT), hypromellose, polyethylene glycol 400, polyethylene oxide, magnesium stearate, titanium dioxide. 10 mg: Hydroxypropyl cellulose. 20 mg: Polysorbate 80 and red iron oxide. 40 mg: Polysorbate 80 and yellow iron oxide.
Action
Pharmacology: Oxycodone is a full opiod agonist and is relatively selective for the mu receptor although it can bind to other opioid receptors at higher doses. The principal therapeutic action of oxycodone is analgesic. Like all full opioid agonists, there is no ceiling effect to analgesia for Oxycodone. Clinically, dosage is titrated to provide adequate analgesia and may be limited by adverse reactions, including respiratory and CNS depression. Mechanism of Action: Central Nervous System: The precise mechanism of the analgesic action is unknown. However, specific CNS opioid receptors for endogenous compounds with opioid-like activity have been identified throughout the brain and spinal cord and are thought to play a role in the analgesic effects of this drug. Pharmacodynamics: A single-dose, double-blind, placebo- and dose-controlled study was conducted using OxyContin (10, 20, and 30 mg) in an analgesic pain model involving 182 patients with moderate to severe pain. OxyContin doses of 20 mg and 30 mg produced statistically significant pain reduction compared to placebo. Effects on the Central Nervous System: Oxycodone produces respiratory depression by direct action on brain stem respiratory centers. The respiratory depression involves both a reduction in the responsiveness of the brain stem respiratory centers to increases in CO2 tension and to electrical stimulation. Oxycodone depresses the cough reflex by direct effect on the cough center in the medulla. Antitussive effects may occur with doses lower than those usually required for analgesia. Oxycodone causes miosis, even in total darkness. Pinpoint pupils are a sign of opioid overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origin may produce similar findings). Marked mydriasis rather than miosis may be seen with hypoxia in the setting of oxycodone overdose (see Overdosage). Effects on the Gastrointestinal Tract and Other Smooth Muscle: Oxycodone causes a reduction in motility associated with an increase in smooth muscle tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone may be increased to the point of spasm resulting in constipation. Other opioid-induced effects may include a reduction in gastric, biliary and pancreatic secretions, spasm of sphincter of Oddi, and transient elevations in serum amylase. Effects on the Cardiovascular System: Oxycodone may produce release of histamine with or without associated peripheral vasodilation. Manifestations of histamine release and/or peripheral vasodilation may include pruritus, flushing, red eyes, sweating, and/or orthostatic hypotension. Effects on the Endocrine System: Opioids inhibit the secretion of ACTH, cortisol, testosterone, and luteinizing hormone (LH) in humans. They also stimulate prolactin, growth hormone (GH) secretion, and pancreatic secretion of insulin and glucagon. Effects on the Immune System: Opioids have been shown to have a variety of effects on components of the immune system in in vitro and animal models. The clinical significance of these findings is unknown. Overall, the effects of opioids appear to be modestly immunosuppressive. Concentration-Efficacy Relationships: Studies in normal volunteers and patients reveal predictable relationships between oxycodone dosage and plasma oxycodone concentrations, as well as between concentration and certain expected opioid effects, such as pupillary constriction, sedation, overall subjective "drug effect", analgesia and feelings of relaxation. The minimum effective analgesic concentration will vary widely among patients, especially among patients who have been previously treated with potent agonist opioids. As a result, patients must be treated with individualized titration of dosage to the desired effect. The minimum effective analgesic concentration of oxycodone for any individual patient may increase over time due to an increase in pain, the development of a new pain syndrome and/or the development of analgesic tolerance. Concentration-Adverse Reaction Relationships: There is a relationship between increasing oxycodone plasma concentration and increasing frequency of dose-related opioid adverse reactions such as nausea, vomiting, CNS effects, and respiratory depression. In opioid-tolerant patients, the situation may be altered by the development of tolerance to opioid-related side effects. The dose of OxyContin Neo must be individualized because the effective analgesic dose for some patients will be too high to be tolerated by other patients (see Dosage and Administration). Clinical Studies: A double-blind, placebo-controlled, fixed-dose, parallel group, two-week study was conducted in 133 patients with persistent, moderate to severe pain, who were judged as having inadequate pain control with their current therapy. In this study, OxyContin 20 mg, but not 10 mg, was statistically significant in pain reduction compared with placebo. Pharmacokinetics: The activity of OxyContin Neo is primarily due to the parent drug oxycodone. OxyContin Neo is designed to provide delivery of oxycodone over 12 hours. Cutting, breaking, chewing, crushing or dissolving OxyContin Neo impairs the controlled-release delivery mechanism and results in the rapid release and absorption of a potentially fatal dose of oxycodone. Oxycodone release from OxyContin Neo is pH independent. The oral bioavailability of oxycodone is 60% to 87%. The relative oral bioavailability of oxycodone from OxyContin Neo to that from immediate-release oral dosage forms is 100%. Upon repeated dosing with OxyContin Neo in healthy subjects in pharmacokinetic studies, steady-state levels were achieved within 24-36 hours. Oxycodone is extensively metabolized and eliminated primarily in the urine as both conjugated and unconjugated metabolites. The apparent elimination half-life (t½) of oxycodone following the administration of OxyContin Neo was 4.5 hours compared to 3.2 hours for immediate-release oxycodone. Absorption: About 60% to 87% of an oral dose of oxycodone reaches the central compartment in comparison to a parenteral dose. This high oral bioavailability is due to low pre-systemic and/or first-pass metabolism. Plasma Oxycodone Concentration over Time: Dose proportionality has been established for OxyContin Neo 10 mg, 15 mg, 20 mg, 30 mg, 40 mg, 60 mg, and 80 mg tablet strengths for both peak plasma concentrations (Cmax) and extent of absorption (AUC) (see Table 1). Given the short elimination t½ of oxycodone, steady-state plasma concentrations of oxycodone are achieved within 24-36 hours of initiation of dosing with OxyContin Neo. In a study comparing 10 mg of OxyContin Neo every 12 hours to 5 mg of immediate-release oxycodone every 6 hours, the two treatments were found to be equivalent for AUC and Cmax, and similar for Cmin (trough) concentrations.
Food Effects: Food has no significant effect on the extent of absorption of oxycodone from OxyContin Neo.
Distribution: Following intravenous administration, the steady-state volume of distribution (Vss) for oxycodone was 2.6 L/kg. Oxycodone binding to plasma protein at 37°C and a pH of 7.4 was about 45%. Once absorbed, oxycodone is distributed to skeletal muscle, liver, intestinal tract, lungs, spleen, and brain. Oxycodone has been found in breast milk (see Use in Specific Populations). Metabolism: Oxycodone is extensively metabolized by multiple metabolic pathways to produce noroxycodone, oxymorphone and noroxymorphone, which are subsequently glucuronidated. Noroxycodone and noroxymorphone are the major circulating metabolites. CYP3A mediated N-demethylation to noroxycodone is the primary metabolic pathway of oxycodone with a lower contribution from CYP2D6 mediated O-demethylation to oxymorphone. Therefore, the formation of these and related metabolites can, in theory, be affected by other drugs (see Interactions). Noroxycodone exhibits very weak anti-nociceptive potency compared to oxycodone, however, it undergoes further oxidation to produce noroxymorphone, which is active at opioid receptors. Although noroxymorphone is an active metabolite and present at relatively high concentrations in circulation, it does not appear to cross the blood-brain barrier to a significant extent. Oxymorphone is present in the plasma only at low concentrations and undergoes further metabolism to form its glucuronide and noroxymorphone. Oxymorphone has been shown to be active and possessing analgesic activity but its contribution to analgesia following oxycodone administration is thought to be clinically insignificant. Other metabolites (α- and ß-oxycodol, noroxycodol and oxymorphol) may be present at very low concentrations and demonstrate limited penetration into the brain as compared to oxycodone. The enzymes responsible for keto-reduction and glucuronidation pathways in oxycodone metabolism have not been established. Excretion: Oxycodone and its metabolites are excreted primarily via the kidney. The amounts measured in the urine have been reported as follows: free and conjugated oxycodone 8.9%, free noroxycodone 23%, free oxymorphone less than 1%, conjugated oxymorphone 10%, free and conjugated noroxymorphone 14%, reduced free and conjugated metabolites up to 18%. The total plasma clearance was approximately 1.4 L/min in adults. Special Populations: Elderly (≥ 65 years): The plasma concentrations of oxycodone are only nominally affected by age, being 15% greater in elderly as compared to young subjects (age 21-45). Gender: Across individual pharmacokinetic studies, average plasma oxycodone concentrations for female subjects were up to 25% higher than for male subjects on a body weight-adjusted basis. The reason for this difference is unknown (see Precautions). Renal Impairment: Data from a pharmacokinetic study involving 13 patients with mild to severe renal dysfunction (creatinine clearance <60 mL/min) showed peak plasma oxycodone and noroxycodone concentrations 50% and 20% higher, respectively, and AUC values for oxycodone, noroxycodone, and oxymorphone 60%, 50%, and 40% higher than normal subjects, respectively. This was accompanied by an increase in sedation but not by differences in respiratory rate, pupillary constriction, or several other measures of drug effect. There was an increase in mean elimination t½ for oxycodone of 1 hour. Hepatic Impairment: Data from a study involving 24 patients with mild to moderate hepatic dysfunction show peak plasma oxycodone and noroxycodone concentrations 50% and 20% higher, respectively, than healthy subjects. AUC values are 95% and 65% higher, respectively. Oxymorphone peak plasma concentrations and AUC values are lower by 30% and 40%. These differences are accompanied by increases in some, but not other, drug effects. The mean elimination t½ for oxycodone increased by 2.3 hours. Drug-Drug Interactions: CYP3A4 Inhibitors: CYP3A4 is the major enzyme involved in noroxycodone formation. Co-administration of OxyContin Neo (10 mg single dose) and the CYP3A4 inhibitor ketoconazole (200 mg BID) increased oxycodone AUC and Cmax by 170% and 100%, respectively (see Interactions). CYP3A4 Inducers: A published study showed that the co-administration of rifampin, a drug metabolizing enzyme inducer, decreased oxycodone AUC and Cmax values by 86% and 63%, respectively (see Interactions). CYP2D6 Inhibitors: Oxycodone is metabolized in part to oxymorphone via CYP2D6. While this pathway may be blocked by a variety of drugs such as certain cardiovascular drugs (e.g., quinidine) and antidepressants (e.g., fluoxetine), such blockade has not been shown to be of clinical significance with OxyContin Neo (see Interactions). Toxicology: Carcinogenesis: No animal studies to evaluate the carcinogenic potential of oxycodone have been conducted. Mutagenesis: Oxycodone was genotoxic in the mouse lymphoma assay at concentrations of 50 mcg/mL or greater with metabolic activation and at 400 mcg/mL or greater without metabolic activation. Clastogenicity was observed with oxycodone in the presence of metabolic activation in one chromosomal aberration assay in human lymphocytes at concentrations greater than or equal to 1250 mcg/mL at 24 but not 48 hours of exposure. In a second chromosomal aberration assay with human lymphocytes, no structural clastogenicity was observed either with or without metabolic activation; however, in the absence of metabolic activation, oxycodone increased numerical chromosomal aberrations (polyploidy). Oxycodone was not genotoxic in the following assays: Ames S. typhimurium and E. coli test with and without metabolic activation at concentrations up to 5000 μg/plate, chromosomal aberration test in human lymphocytes (in the absence of metabolic activation) at concentrations up to 1500 μg/mL, and with activation after 48 hours of exposure at concentrations up to 5000 μg/mL, and in the in vivo bone marrow micronucleus assay in mice (at plasma levels up to 48 μg/mL). Impairment of Fertility: In a study of reproductive performance, rats were administered a once daily gavage dose of the vehicle or oxycodone hydrochloride (0.5, 2, and 8 mg/kg). Male rats were dosed for 28 days before cohabitation with females, during the cohabitation and until necropsy (2-3 weeks post-cohabitation). Females were dosed for 14 days before cohabitation with males, during cohabitation and up to gestation day 6. Oxycodone hydrochloride did not affect reproductive function in male or female rats at any dose tested (≤8 mg/kg/day).
Indications/Uses
OxyContin Neo is indicated for the management of pain severe enough to require daily, around the-clock, long-term opioid treatment and for which alternative treatment options are inadequate. Limitations of Use: Because of the risks of addiction, abuse, and misuse with opioids, even at recommended doses, and because of the greater risks of overdose and death with extended-release opioid formulations, reserve OxyContin Neo for use in patients for whom alternative treatment options (e.g., non-opioid analgesics or immediate-release opioids) are ineffective, not tolerated, or would be otherwise inadequate to provide sufficient management of pain. OxyContin Neo is not indicated as an as-needed (prn) analgesic. Dosage/Direction for Use Initial Dosing: OxyContin Neo should be prescribed only by healthcare professionals who are knowledgeable in the use of potent opioids for the management of chronic pain. OxyContin Neo 60 mg and 80 mg tablets, a single dose greater than 40 mg, or a total daily dose greater than 80 mg are only for use in patients in whom tolerance to an opioid of comparable potency has been established. Patients considered opioid tolerant are those receiving, for one week or longer, at least 60 mg oral morphine/day, 25 mcg transdermal fentanyl/hour, 30 mg oral oxycodone/day, 8 mg oral hydromorphone/day, 25 mg oral oxymorphone/day, or an equianalgesic dose of another opioid. Initiate the dosing regimen for each patient individually, taking into account the patient's prior analgesic treatment experience, and risk factors for addiction, abuse, and misuse (see Precautions). Monitor patients closely for respiratory depression, especially within the first 24-72 hours of initiating therapy with OxyContin Neo (see Precautions). OxyContin Neo tablets must be taken whole, one tablet at a time, with enough water to ensure complete swallowing immediately after placing in the mouth. Crushing, chewing, or dissolving OxyContin Neo tablets will result in uncontrolled delivery of oxycodone and can lead to overdose or death (see Precautions). Use of OxyContin Neo as the First Opioid Analgesic: Initiate treatment with OxyContin Neo with one 10 mg tablet orally every 12 hours. Use of OxyContin Neo in Patients who are not Opioid Tolerant: The starting dose for patients who are not opioid tolerant is OxyContin Neo 10 mg orally every 12 hours. Patients who are opioid tolerant are those receiving, for one week or longer, at least 60 mg oral morphine per day, 25 mcg transdermal fentanyl per hour, 30 mg oral oxycodone per day, 8 mg oral hydromorphone per day, 25 mg oral oxymorphone per day, or an equianalgesic dose of another opioid. Use of higher starting doses in patients who are not opioid tolerant may cause fatal respiratory depression. Conversion from other Oral Oxycodone Formulations to OxyContin Neo: Patients receiving other oral oxycodone formulations may be converted to OxyContin Neo by administering one-half of the patient's total daily oral oxycodone dose as OxyContin Neo every 12 hours. It is safer to underestimate a patient's 24-hour oral oxycodone requirements and provide rescue medication (e.g., immediate-release opioid) than to overestimate the 24-hour oral oxycodone requirements which could result in adverse reactions. While useful tables of opioid equivalents are readily available, there is substantial inter-patient variability in the relative potency of different opioid drugs and products. Conversion from Methadone to OxyContin Neo: Close monitoring is of particular importance when converting from methadone to other opioid agonists. The ratio between methadone and other opioid agonists may vary widely as a function of previous dose exposure. Methadone has a long half-life and can accumulate in the plasma. Conversion from Transdermal Fentanyl to OxyContin Neo: Eighteen hours following the removal of the transdermal fentanyl patch, OxyContin Neo treatment can be initiated. Although there has been no systematic assessment of such conversion, a conservative oxycodone dose, approximately 10 mg every 12 hours of OxyContin Neo, should be initially substituted for each 25 mcg/hr fentanyl transdermal patch. Follow the patient closely during conversion from transdermal fentanyl to OxyContin Neo, as there is limited documented experience with this conversion. Titration and Maintenance of Therapy: Individually titrate OxyContin Neo to a dose that provides adequate analgesia and minimizes adverse reactions. Continually reevaluate patients receiving OxyContin Neo to assess the maintenance of pain control and the relative incidence of adverse reactions as well as monitoring for the development of addiction, abuse and misuse. Frequent communication is important among the prescriber, other members of the healthcare team, the patient, and the caregiver/family during periods of changing analgesic requirements, including initial titration. During chronic therapy, periodically reassess the continued need for the use of opioid analgesics. Patients who experience breakthrough pain may require a dose increase of OxyContin Neo or may need rescue medication with an appropriate dose of an immediate-release analgesic. If the level of pain increases after dose stabilization, attempt to identify the source of increased pain before increasing the OxyContin Neo dose. Because steady-state plasma concentrations are approximated in 1 day, OxyContin Neo dosage may be adjusted every 1 to 2 days. If unacceptable opioid-related adverse reactions are observed, the subsequent dose may be reduced. Adjust the dose to obtain an appropriate balance between management of pain and opioid-related adverse reactions. There are no well-controlled clinical studies evaluating the safety and efficacy with dosing more frequently than every 12 hours. As a guideline, the total daily oxycodone dose usually can be increased by 25% to 50% of the current dose, each time an increase is clinically indicated. Patients with Hepatic Impairment: For patients with hepatic impairment, start dosing patients at 1/3 to 1/2 the usual starting dose followed by careful dose titration (see Pharmacology under Actions). Discontinuation of OxyContin Neo: When the patient no longer requires therapy with OxyContin Neo tablets, use a gradual downward titration of the dose to prevent signs and symptoms of withdrawal in the physically-dependent patient. Do not abruptly discontinue OxyContin Neo. Administration: Instruct patients to swallow OxyContin Neo tablets intact. The tablets are not to be crushed, dissolved, or chewed due to the risk of rapid release and absorption of a potentially fatal dose of oxycodone (see Precautions). Instruct patients to take OxyContin Neo one tablet at a time and with enough water to ensure complete swallowing immediately after placing in the mouth (see Precautions).
Overdosage
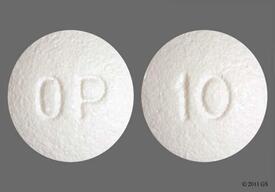
Clinical Presentation: Acute overdosage with OxyContin Neo can be manifested by respiratory depression, somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and in some cases, pulmonary edema, bradycardia, hypotension, partial or complete airway obstruction, atypical snoring and death. Marked mydriasis rather than miosis may be seen due to severe hypoxia in overdose situations. Treatment of Overdose: In case of overdose, priorities are the reestablishment of a patent and protected airway and institution of assisted or controlled ventilation if needed. Employ other supportive measures (including oxygen, vasopressors) in the management of circulatory shock and pulmonary edema as indicated. Cardiac arrest or arrhythmias will require advanced life support techniques. The opioid antagonists, naloxone or nalmefene, are specific antidotes to respiratory depression resulting from opioid overdose. Opioid antagonists should not be administered in the absence of clinically significant respiratory or circulatory depression secondary to oxycodone overdose. Such agents should be administered cautiously to persons who are known, or suspected to be physically dependent on OxyContin Neo. In such cases, an abrupt or complete reversal of opioid effects may precipitate an acute withdrawal syndrome. Because the duration of reversal would be expected to be less than the duration of action of oxycodone in OxyContin Neo, carefully monitor the patient until spontaneous respiration is reliably reestablished. OxyContin Neo will continue to release oxycodone and add to the oxycodone load for 24 to 48 hours or longer following ingestion, necessitating prolonged monitoring. If the response to opioid antagonists is suboptimal or not sustained, additional antagonist should be administered as directed in the product's prescribing information. In an individual physically dependent on opioids, administration of the usual dose of the antagonist will precipitate an acute withdrawal syndrome. The severity of the withdrawal symptoms experienced will depend on the degree of physical dependence and the dose of the antagonist administered. If a decision is made to treat serious respiratory depression in the physically dependent patient, administration of the antagonist should be begun with care and by titration with smaller than usual doses of the antagonist. Contraindications Hypersensitivity to opioids and to any of the constituents or in any situation where opioids are contraindicated, acute respiratory depression, cor pulmonale, cardiac arrhythmias, severe bronchial asthma, chronic obstructive airways disease, paralytic ileus, suspected surgical abdomen, moderate to severe hepatic impairment, severe renal impairment (creatinine clearance <10 ml/min), delayed gastric emptying, acute alcoholism, brain tumor, increased cerebrospinal or intracranial pressure, head injury, severe CNS depression, convulsive disorders, delirium tremens, hypercarbia, concurrent administration of monoamine oxidase inhibitors or within 2 weeks of discontinuation of their use. Not recommended for pre-operative use or for the first 24 hours post-operatively. Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption should not take this medicine. Special Precautions Addiction, Abuse, and Misuse: OxyContin Neo contains oxycodone. As an opioid, OxyContin Neo exposes users to the risks of addiction, abuse, and misuse (see Drug Abuse and Dependence). As modified-release products such as OxyContin Neo deliver the opioid over an extended period of time, there is a greater risk for overdose and death due to the larger amount of oxycodone present (see Drug Abuse and Dependence). Although the risk of addiction in any individual is unknown, it can occur in patients appropriately prescribed OxyContin Neo. Addiction can occur at recommended doses and if the drug is misused or abused. Assess each patient's risk for opioid addiction, abuse or misuse prior to prescribing OxyContin Neo, and monitor all patients receiving OxyContin Neo for the development of these behaviors or conditions. Risks are increased in patients with a personal or family history of substance abuse (including drug or alcohol abuse or addiction) or mental illness (e.g., major depression). The potential for these risks should not, however, prevent the proper management of pain in any given patient. Patients at increased risk may be prescribed modified-release opioid formulations such as OxyContin Neo, but use in such patients necessitates intensive counseling about the risks and proper use of OxyContin Neo along with intensive monitoring for signs of addiction, abuse, and misuse. Abuse, or misuse of OxyContin Neo by crushing, chewing, snorting, or injecting the dissolved product will result in the uncontrolled delivery of oxycodone and can result in overdose and death (see Overdosage). Opioid agonists are sought by drug abusers and people with addiction disorders and are subject to criminal diversion. Consider these risks when prescribing or dispensing OxyContin Neo. Strategies to reduce these risks include prescribing the drug in the smallest appropriate quantity and advising the patient on the proper disposal of unused drug. Life-Threatening Respiratory Depression: Serious, life-threatening, or fatal respiratory depression has been reported with the use modified-release opioids, even when used as recommended. Respiratory depression, if not immediately recognized and treated, may lead to respiratory arrest and death. Management of respiratory depression may include close observation, supportive measures, and use of opioid antagonists, depending on the patient’s clinical status (see Overdosage). Carbon dioxide (CO2) retention from opioid-induced respiratory depression can exacerbate the sedating effects of opioids. While serious, life-threatening, or fatal respiratory depression can occur at any time during the use of OxyContin Neo, the risk is greatest during the initiation of therapy or following a dose increase. Closely monitor patients for respiratory depression when initiating therapy with OxyContin Neo and following dose increases. To reduce the risk of respiratory depression, proper dosing and titration of OxyContin Neo are essential (see Dosage and Administration). Overestimating the OxyContin Neo dose when converting patients from another opioid product can result in a fatal overdose with the first dose. Accidental ingestion of even one dose of OxyContin Neo, especially by children, can result in respiratory depression and death due to an overdose of oxycodone. Neonatal Opioid Withdrawal Syndrome: Prolonged use of OxyContin Neo during pregnancy can result in withdrawal signs in the neonate. Neonatal opioid withdrawal syndrome, unlike opioid withdrawal syndrome in adults, may be life-threatening if not recognized and treated, and requires management according to protocols developed by neonatology experts. If opioid use is required for a prolonged period in a pregnant woman, advise the patient of the risk of neonatal opioid withdrawal syndrome and ensure that appropriate treatment will be available. Neonatal opioid withdrawal syndrome presents as irritability, hyperactivity and abnormal sleep pattern, high pitched cry, tremor, vomiting, diarrhea and failure to gain weight. The onset, duration, and severity of neonatal opioid withdrawal syndrome vary based on the specific opioid used, duration of use, timing and amount of last maternal use, and rate of elimination of the drug by the newborn. Interactions with Central Nervous System Depressants: Hypotension and profound sedation, coma, or respiratory depression may result if OxyContin Neo is used concomitantly with other central nervous system (CNS) depressants (e.g., sedatives, anxiolytics, hypnotics, neuroleptics, other opioids).When considering the use of OxyContin Neo in a patient taking a CNS depressant, assess the duration of use of the CNS depressant and the patient’s response, including the degree of tolerance that has developed to CNS depression. Additionally, evaluate the patient's use of alcohol or illicit drugs that can cause CNS depression. If the decision to begin OxyContin Neo therapy is made, start with 1/3 to 1/2 the usual dose of OxyContin Neo, monitor patients for signs of sedation and respiratory depression and consider using a lower dose of the concomitant CNS depressant (see Interactions and Dosage & Administration). When considering the use of OxyContin Neo in a patient taking a CNS depressant, assess the duration of use of the CNS depressant and the patient’s response, including the degree of tolerance that has developed to CNS depression. Additionally, evaluate the patient's use of alcohol or illicit drugs that can cause CNS depression. If the decision to begin OxyContin Neo therapy is made, start with 1/3 to 1/2 the usual dose of OxyContin Neo, monitor patients for signs of sedation and respiratory depression and consider using a lower dose of the concomitant CNS depressant (see Drug Interactions and Dosage & Administration). Elderly, Cachectic, and Debilitated Patients: Respiratory depression is more likely to occur in elderly, cachectic, or debilitated patients as they may have altered pharmacokinetics or altered clearance compared to younger, healthier patients. Therefore, monitor such patients closely, particularly when initiating and titrating OxyContin Neo and when OxyContin Neo is given concomitantly with other drugs that depress respiration (see Precautions). Use in Patients with Chronic Pulmonary Disease: Monitor patients with significant chronic obstructive pulmonary disease or cor pulmonale, and patients having a substantially decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression for respiratory depression, particularly when initiating therapy and titrating with OxyContin Neo, as in these patients, even usual therapeutic doses of OxyContin Neo may decrease respiratory drive to the point of apnea (see Precautions). Consider the use of alternative non-opioid analgesics in these patients if possible. Hypotensive Effects: OxyContin Neo may cause severe hypotension including orthostatic hypotension and syncope in ambulatory patients. There is an increased risk in patients whose ability to maintain blood pressure has already been compromised by a reduced blood volume or concurrent administration of certain CNS depressant drugs (e.g., phenothiazines or general anesthetics) (see Interactions). Monitor these patients for signs of hypotension after initiating or titrating the dose of OxyContin Neo. In patients with circulatory shock, OxyContin Neo may cause vasodilation that can further reduce cardiac output and blood pressure. Avoid the use of OxyContin Neo in patients with circulatory shock. Use in Patients with Head Injury or Increased Intracranial Pressure: Monitor patients taking OxyContin Neo who may be susceptible to the intracranial effects of CO2 retention (e.g., those with evidence of increased intracranial pressure or brain tumors) for signs of sedation and respiratory depression, particularly when initiating therapy with OxyContin Neo. OxyContin Neo may reduce respiratory drive, and the resultant CO2 retention can further increase intracranial pressure. Opioids may also obscure the clinical course in a patient with a head injury. Avoid the use of OxyContin Neo in patients with impaired consciousness or coma. Difficulty in Swallowing and Risk for Obstruction in Patients at Risk for a Small Gastrointestinal Lumen: There have been post-marketing reports of difficulty in swallowing OxyContin Neo tablets. These reports included choking, gagging, regurgitation and tablets stuck in the throat. Instruct patients not to pre-soak, lick or otherwise wet OxyContin Neo tablets prior to placing in the mouth, and to take one tablet at a time with enough water to ensure complete swallowing immediately after placing in the mouth. There have been rare post-marketing reports of cases of intestinal obstruction, and exacerbation of diverticulitis, some of which have required medical intervention to remove the tablet. Patients with underlying GI disorders such as esophageal cancer or colon cancer with a small gastrointestinal lumen are at greater risk of developing these complications. Consider use of an alternative analgesic in patients who have difficulty swallowing and patients at risk for underlying GI disorders resulting in a small gastrointestinal lumen. Use in Patients with Gastrointestinal Conditions: OxyContin Neo is contraindicated in patients with GI obstruction, including paralytic ileus. The oxycodone in OxyContin Neo may cause spasm of the sphincter of Oddi. Monitor patients with biliary tract disease, including acute pancreatitis, for worsening symptoms. Opioids may cause increases in the serum amylase. Use in Patients with Convulsive or Seizure Disorders: The oxycodone in OxyContin Neo may aggravate convulsions in patients with convulsive disorders, and may induce or aggravate seizures in some clinical settings. Monitor patients with a history of seizure disorders for worsened seizure control during OxyContin Neo therapy. Avoidance of Withdrawal: Avoid the use of mixed agonist/antagonist analgesics (i.e., pentazocine, nalbuphine, and butorphanol) in patients who have received or are receiving a course of therapy with a full opioid agonist analgesic, including OxyContin Neo. In these patients, mixed agonist/antagonist analgesics may reduce the analgesic effect and/or may precipitate withdrawal symptoms. When discontinuing OxyContin Neo, gradually taper the dose (see Dosage and Administration). Do not abruptly discontinue OxyContin Neo. Driving and Operating Machinery: OxyContin Neo may impair the mental or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery. Warn patients not to drive or operate dangerous machinery unless they are tolerant to the effects of OxyContin Neo and know how they will react to the medication. Cytochrome P450 3A4 Inhibitors and Inducers: Since the CYP3A4 isoenzyme plays a major role in the metabolism of OxyContin Neo, drugs that alter CYP3A4 activity may cause changes in clearance of oxycodone which could lead to changes in oxycodone plasma concentrations. Inhibition of CYP3A4 activity by its inhibitors, such as macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), and protease inhibitors (e.g., ritonavir), may increase plasma concentrations of oxycodone and prolong opioid effects. CYP450 inducers, such as rifampin, carbamazepine, and phenytoin, may induce the metabolism of oxycodone and, therefore, may cause increased clearance of the drug which could lead to a decrease in oxycodone plasma concentrations, lack of efficacy or, possibly, development of an abstinence syndrome in a patient who had developed physical dependence to oxycodone. If co-administration is necessary, caution is advised when initiating OxyContin Neo treatment in patients currently taking, or discontinuing, CYP3A4 inhibitors or inducers. Evaluate these patients at frequent intervals and consider dose adjustments until stable drug effects are achieved (see Interactions and Pharmacology under Actions). Laboratory Monitoring: Not every urine drug test for "opioids" or "opiates" detects oxycodone reliably, especially those designed for in-office use. Further, many laboratories will report urine drug concentrations below a specified "cut-off" value as "negative". Therefore, if urine testing for oxycodone is considered in the clinical management of an individual patient, ensure that the sensitivity and specificity of the assay is appropriate, and consider the limitations of the testing used when interpreting results. Gender Differences: In pharmacokinetic studies with OxyContin Neo, opioid-naïve females demonstrate up to 25% higher average plasma concentrations and greater frequency of typical opioid adverse events than males, even after adjustment for body weight. The clinical relevance of a difference of this magnitude is low for a drug intended for chronic usage at individualized dosages, and there was no male/female difference detected for efficacy or adverse events in clinical trials. Neonatal Opioid Withdrawal Syndrome: Chronic maternal use of oxycodone during pregnancy can affect the fetus with subsequent withdrawal signs. Neonatal withdrawal syndrome presents as irritability, hyperactivity and abnormal sleep pattern, high pitched cry, tremor, vomiting, diarrhea and failure to gain weight. The onset, duration and severity of neonatal withdrawal syndrome vary based on the drug used, duration of use, the dose of last maternal use, and rate of elimination of drug by the newborn. Neonatal opioid withdrawal syndrome, unlike opioid withdrawal syndrome in adults, may be life-threatening and should be treated according to protocols developed by neonatology experts. Hepatic Impairment: A study of OxyContin Neo in patients with hepatic impairment demonstrated greater plasma concentrations than those seen at equivalent doses in persons with normal hepatic function. Therefore, in the setting of hepatic impairment, start dosing patients at 1/3 to 1/2 the usual starting dose followed by careful dose titration (see Pharmacology under Actions). Renal Impairment: In patients with renal impairment, as evidenced by decreased creatinine clearance (<60 mL/min), the concentrations of oxycodone in the plasma are approximately 50% higher than in subjects with normal renal function. Follow a conservative approach to dose initiation and adjust according to the clinical situation (see Pharmacology under Actions). Drug Abuse and Dependence: Opioids: OxyContin Neo contains oxycodone, an opioid with a high potential for abuse similar to other opioids including fentanyl, hydromorphone, methadone, oxycodone, and oxymorphone. OxyContin Neo can be abused and is subject to misuse, addiction, and criminal diversion (see Precautions). The high drug content in extended-release formulations adds to the risk of adverse outcomes from abuse and misuse. Abuse: All patients treated with opioids require careful monitoring for signs of abuse and addiction, since use of opioid analgesic products carries the risk of addiction even under appropriate medical use. Drug abuse is the intentional non-therapeutic use of an over-the-counter or prescription drug, even once, for its rewarding psychological or physiological effects. Drug abuse includes, but is not limited to, the following examples: The use of a prescription or over-the-counter drug to get "high", or the use of steroids for performance enhancement and muscle build up. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that develop after repeated substance use and include: a strong desire to take the drug, difficulties in controlling its use, persisting in its use despite harmful consequences, a higher priority given to drug use than to other activities and obligations, increased tolerance, and sometimes a physical withdrawal. "Drug-seeking" behavior is very common to addicts and drug abusers. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing or referral, repeated claims of loss of prescriptions, tampering with prescriptions and reluctance to provide prior medical records or contact information for other treating physician(s). "Doctor shopping" (visiting multiple prescribers) to obtain additional prescriptions is common among drug abusers and people suffering from untreated addiction. Preoccupation with achieving adequate pain relief can be appropriate behavior in a patient with poor pain control. Abuse and addiction are separate and distinct from physical dependence and tolerance. Physicians should be aware that addiction may not be accompanied by concurrent tolerance and symptoms of physical dependence in all addicts. In addition, abuse of opioids can occur in the absence of true addiction. OxyContin Neo, like other opioids, can be diverted for non-medical use into illicit channels of distribution. Careful recordkeeping of prescribing information, including quantity, frequency, and renewal requests as required by state law, is strongly advised. Proper assessment of the patient, proper prescribing practices, periodic reevaluation of therapy, and proper dispensing and storage are appropriate measures that help to reduce abuse of opioid drugs. Risks Specific to Abuse of OxyContin Neo: OxyContin Neo is for oral use only. Abuse of OxyContin Neo poses a risk of overdose and death. The risk is increased with concurrent use of OxyContin Neo with alcohol and other central nervous system depressants. Taking cut, broken, chewed, crushed, or dissolved OxyContin Neo enhances drug release and increases the risk of overdose and death. With parenteral abuse, the inactive ingredients in OxyContin Neo can result in death, local tissue necrosis, infection, pulmonary granulomas, and increased risk of endocarditis and valvular heart injury. Parenteral drug abuse is commonly associated with transmission of infectious diseases, such as hepatitis and HIV. Abuse Deterrence Studies: OxyContin Neo is formulated with inactive ingredients intended to make the tablet more difficult to manipulate for misuse and abuse. For the purposes of describing the results of studies of the abuse-deterrent characteristics of OxyContin Neo resulting from a change in formulation, in this section, the original formulation of OxyContin, which is no longer marketed, will be referred to as "original OxyContin" and the reformulated, currently marketed product will be referred to as OxyContin Neo. In Vitro Testing: In vitro physical and chemical tablet manipulation studies were performed to evaluate the success of different extraction methods in defeating the extended-release formulation. Results support that, relative to original OxyContin Neo, there is an increase in the ability of OxyContin Neo to resist crushing, breaking, and dissolution using a variety of tools and solvents. The results of these studies also support this finding for OxyContin Neo relative to an immediate-release oxycodone. When subjected to an aqueous environment, OxyContin Neo gradually forms a viscous hydrogel (i.e., a gelatinous mass) that resists passage through a needle. Clinical Studies: In a randomized, double-blind, placebo-controlled 5-period crossover pharmacodynamic study, 30 recreational opioid users with a history of intranasal drug abuse received intranasally administered active and placebo drug treatments. The five treatment arms were finely crushed OxyContin Neo 30 mg tablets, coarsely crushed OxyContin Neo 30 mg tablets, finely crushed original OxyContin Neo 30 mg tablets, powdered oxycodone HCl 30 mg, and placebo. Data for finely crushed OxyContin Neo, finely crushed original OxyContin Neo, and powdered oxycodone HCl are described as follows. Drug liking was measured on a bipolar drug liking scale of 0 to 100 where 50 represents a neutral response of neither liking nor disliking, 0 represents maximum disliking and 100 represents maximum liking. Response to whether the subject would take the study drug again was also measured on a bipolar scale of 0 to 100 where 50 represents a neutral response, 0 represents the strongest negative response ("definitely would not take drug again") and 100 represents the strongest positive response ("definitely would take drug again"). Twenty-seven of the subjects completed the study. Incomplete dosing due to granules falling from the subjects’ nostrils occurred in 34% (n=10) of subjects with finely crushed OxyContin Neo, compared with 7% (n=2) of subjects with finely crushed original OxyContin and no subjects with powdered oxycodone HCl. The intranasal administration of finely crushed OxyContin Neo was associated with a numerically lower mean and median drug liking score and a lower mean and median score for take drug again, compared to finely crushed original OxyContin or powdered oxycodone HCl as summarized in Table 2. (See Table 2.) Figure as follows demonstrates a comparison of drug liking for finely crushed OxyContin Neo compared to powdered oxycodone HCl in subjects who received both treatments. The Y-axis represents the percent of subjects attaining a percent reduction in drug liking for OxyContin Neo vs. oxycodone HCl powder greater than or equal to the value on the X-axis. Approximately 44% (n=12) had no reduction in liking with OxyContin Neo relative to oxycodoneHCl. Approximately 56% (n=15) of subjects had some reduction in drug liking with OxyContin Neo relative to oxycodone HCl. Thirty-three percent (n=9) of subjects had a reduction of at least 30% in drug liking with OxyContin Neo compared to oxycodone HCl, and approximately 22% (n=6) of subjects had a reduction of at least 50% in drug liking with OxyContin Neo compared to oxycodone HCl (see Figure.) The results of a similar analysis of drug liking for finely crushed OxyContin Neo relative to finely crushed original OxyContin were comparable to the results of finely crushed OxyContin Neo relative to powdered oxycodone HCl. Approximately 43% (n=12) of subjects had no reduction in liking with OxyContin Neo relative to original OxyContin. Approximately 57% (n=16) of subjects had some reduction in drug liking, 36% (n=10) of subjects had a reduction of at least 30% in drug liking, and approximately 29% (n=8) of subjects had a reduction of at least 50% in drug liking with OxyContin Neo compared to original OxyContin. Summary: The in vitro data demonstrate that OxyContin Neo has physicochemical properties expected to make abuse via injection difficult. The data from the clinical study, along with support from the in vitro data, also indicate that OxyContin Neo has physicochemical properties that are expected to reduce abuse via the intranasal route. However, abuse of OxyContin Neo by these routes, as well as by the oral route is still possible. Additional data, including epidemiological data, when available, may provide further information on the impact of the current formulation of OxyContin Neo on the abuse liability of the drug. Accordingly, this section may be updated in the future as appropriate. OxyContin Neo contains oxycodone, an opioid agonist with an abuse liability similar to other opioid agonists, legal or illicit, including fentanyl, hydromorphone, methadone, morphine, and oxymorphone. OxyContin Neo can be abused and is subject to misuse, addiction, and criminal diversion (see Precautions). Dependence: Both tolerance and physical dependence can develop during chronic opioid therapy. Tolerance is the need for increasing doses of opioids to maintain a defined effect such as analgesia (in the absence of disease progression or other external factors). Tolerance may occur to both the desired and undesired effects of drugs, and may develop at different rates for different effects. Physical dependence results in withdrawal symptoms after abrupt discontinuation or a significant dose reduction of a drug. Withdrawal also may be precipitated through the administration of drugs with opioid antagonist activity, e.g., naloxone, nalmefene, or mixed agonist/antagonist analgesics (pentazocine, butorphanol, nalbuphine). Physical dependence may not occur to a clinically significant degree until after several days to weeks of continued opioid usage. OxyContin Neo should not be abruptly discontinued (see Dosage and Administration). If OxyContin Neo is abruptly discontinued in a physically-dependent patient, an abstinence syndrome may occur. Some or all of the following can characterize this syndrome: restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other signs and symptoms also may develop, including: irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate. Infants born to mothers physically dependent on opioids will also be physically dependent and may exhibit respiratory difficulties and withdrawal signs. Use in Children: Safety and effectiveness of OxyContin Neo in pediatric patients below the age of 18 years have not been established. Use in Elderly: In controlled pharmacokinetic studies in elderly subjects (greater than 65 years) the clearance of oxycodone was slightly reduced. Compared to young adults, the plasma concentrations of oxycodone were increased approximately 15% (see Pharmacology under Actions). Of the total number of subjects (445) in clinical studies of oxycodone hydrochloride controlled-release tablets, 148 (33.3%) were age 65 and older (including those age 75 and older) while 40 (9.0%) were age 75 and older. In clinical trials with appropriate initiation of therapy and dose titration, no untoward or unexpected adverse reactions were seen in the elderly patients who received oxycodone hydrochloride controlled-release tablets. Thus, the usual doses and dosing intervals may be appropriate for elderly patients. However, reduce the starting dose to 1/3 to 1/2 the usual dosage in debilitated, non-opioid-tolerant patients. Respiratory depression is the chief risk in elderly or debilitated patients, usually the result of large initial doses in patients who are not tolerant to opioids, or when opioids are given in conjunction with other agents that depress respiration. Titrate the dose of OxyContin Neo cautiously in these patients. Use In Pregnancy & Lactation Use in Pregnancy: Clinical Considerations: Fetal/neonatal Adverse Reactions: Prolonged use of opioid analgesics during pregnancy for medical or nonmedical purposes can result in physical dependence in the neonate and neonatal opioid withdrawal syndrome shortly after birth. Observe newborns for symptoms of neonatal opioid withdrawal syndrome, such as poor feeding, diarrhea, irritability, tremor, rigidity, and seizures, and manage accordingly (see Precautions). Teratogenic Effects-Pregnancy Category C: There are no adequate and well-controlled studies in pregnant women. OxyContin Neo should be used during pregnancy only if the potential benefit justifies the risk to the fetus. The effect of oxycodone in human reproduction has not been adequately studied. Studies with oral doses of oxycodone hydrochloride in rats up to 8 mg/kg/day and rabbits up to 125 mg/kg/day, equivalent to 0.5 and 2.0 times an adult human dose of 160 mg/day, respectively on a mg/m2 basis, did not reveal evidence of harm to the fetus due to oxycodone. In a pre- and postnatal toxicity study, female rats received oxycodone during gestation and lactation. There were no long-term developmental or reproductive effects in the pups (see Toxicology under Actions). Non-Teratogenic Effects: Oxycodone hydrochloride was administered orally to female rats during gestation and lactation in a pre- and postnatal toxicity study. There were no drug-related effects on reproductive performance in these females or any long-term developmental or reproductive effects in pups born to these rats. Decreased body weight was found during lactation and the early post-weaning phase in pups nursed by mothers given the highest dose used (6 mg/kg/day, equivalent to approximately 0.4-times an adult human dose of 160 mg/day, on a mg/m2 basis). However, body weight of these pups recovered. Labor and Delivery: Opioids cross the placenta and may produce respiratory depression and psycho-physiologic effects in neonates. OxyContin Neo is not recommended for use in women immediately prior to and during labor, when use of shorter-acting analgesics or other analgesic techniques are more appropriate. Occasionally, opioid analgesics may prolong labor through actions which temporarily reduce the strength, duration and frequency of uterine contractions. However this effect is not consistent and may be offset by an increased rate of cervical dilatation, which tends to shorten labor. Use in Lactation: Oxycodone has been detected in breast milk. Instruct patients not to undertake nursing while receiving OxyContin Neo. Do not initiate OxyContin Neo therapy while nursing because of the possibility of sedation or respiratory depression in the infant. Withdrawal signs can occur in breast-fed infants when maternal administration of an opioid analgesic is stopped, or when breast-feeding is stopped. Adverse Reactions Clinical Trial Experience: Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The safety of OxyContin Neo was evaluated in double-blind clinical trials involving 713 patients with moderate to severe pain of various etiologies. In open-label studies of cancer pain, 187 patients received OxyContin Neo in total daily doses ranging from 20 mg to 640 mg per day. The average total daily dose was approximately 105 mg per day. OxyContin Neo may increase the risk of serious adverse reactions such as those observed with other opioid analgesics, including respiratory depression, apnea, respiratory arrest, circulatory depression, hypotension, or shock (see Overdosage). The most common adverse reactions (>5%) reported by patients in clinical trials comparing OxyContin Neo with placebo are shown in Table 3 as follows. (See Table 3.) In clinical trials, the following adverse reactions were reported in patients treated with OxyContin with an incidence between 1% and 5%: Gastrointestinal Disorders: Abdominal pain, diarrhea, dyspepsia, gastritis. General Disorders and Administration Site Conditions: Chills, fever. Metabolism and Nutrition Disorders: Anorexia. Musculoskeletal and Connective Tissue Disorders: Twitching. Psychiatric Disorders: Abnormal dreams, anxiety, confusion, dysphoria, euphoria, insomnia, nervousness, thought abnormalities. Respiratory, Thoracic and Mediastinal Disorders: Dyspnea, hiccups. Skin and Subcutaneous Tissue Disorders: Rash. Vascular Disorders: Postural hypotension. The following adverse reactions occurred in less than 1% of patients involved in clinical trials: Blood and Lymphatic System Disorders: Lymphadenopathy. Ear and Labyrinth Disorders: Tinnitus. Eye Disorders: Abnormal vision. Gastrointestinal Disorders: Dysphagia, eructation, flatulence, gastrointestinal disorder, increased appetite, stomatitis. General Disorders and Administration Site Conditions: Withdrawal syndrome (with and without seizures), edema, peripheral edema, thirst, malaise, chest pain, facial edema. Injury, Poisoning and Procedural Complications: Accidental injury. Investigations: ST depression. Metabolism and Nutrition Disorders: Dehydration. Nervous System Disorders: Syncope, migraine, abnormal gait, amnesia, hyperkinesia, hypesthesia, hypotonia, paresthesia, speech disorder, stupor, tremor, vertigo, taste perversion. Psychiatric Disorders: Depression, agitation, depersonalization, emotional lability, hallucination. Renal and Urinary Disorders: Dysuria, hematuria, polyuria, urinary retention. Reproductive System and Breast Disorders: Impotence. Respiratory, Thoracic and Mediastinal Disorders: Cough increased, voice alteration. Skin and Subcutaneous Tissue Disorders: Dry skin, exfoliative dermatitis. Postmarketing Experience: The following adverse reactions have been identified during post-approval use of controlled-release oxycodone: abuse, addiction, amenorrhea, cholestasis, death, dental caries, increased hepatic enzymes, hyperalgesia, hyponatremia, ileus, muscular hypertonia, overdose, palpitations (in the context of withdrawal), seizures, syndrome of inappropriate antidiuretic hormone secretion, and urticaria. Anaphylaxis has been reported with ingredients contained in OxyContin Neo. Advise patients how to recognize such a reaction and when to seek medical attention. In addition to the events listed above, the following have also been reported, potentially due to the swelling and hydrogelling property of the tablet: choking, gagging, regurgitation, tablets stuck in the throat and difficulty swallowing the tablet. View ADR Monitoring Form Drug Interactions CNS Depressants: The concomitant use of OxyContin Neo and other CNS depressants including sedatives, hypnotics, tranquilizers, general anesthetics, phenothiazines, other opioids, and alcohol can increase the risk of respiratory depression, profound sedation, coma, or death. Monitor patients receiving CNS depressants and OxyContin Neo for signs of respiratory depression, sedation, and hypotension. When combined therapy with any of the above medications is considered, the dose of one or both agents should be reduced (see Dosage & Administration and Precautions). Muscle Relaxants: Oxycodone may enhance the neuromuscular blocking action of true skeletal muscle relaxants and produce an increased degree of respiratory depression. Monitor patients receiving muscle relaxants and OxyContin Neo for signs of respiratory depression that may be greater than otherwise expected. Agents Affecting Cytochrome P450 Isoenzymes: Inhibitors of CYP3A4 and 2D6: Because the CYP3A4 isoenzyme plays a major role in the metabolism of oxycodone, drugs that inhibit CYP3A4 activity may cause decreased clearance of oxycodone which could lead to an increase in oxycodone plasma concentrations and result in increased or prolonged opioid effects. These effects could be more pronounced with concomitant use of CYP2D6 and 3A4 inhibitors. If co-administration with OxyContin Neo is necessary, monitor patients for respiratory depression and sedation at frequent intervals and consider dose adjustments until stable drug effects are achieved (see Pharmacology under Actions). Inducers of CYP3A4: CYP450 3A4 inducers may induce the metabolism of oxycodone and, therefore, may cause increased clearance of the drug which could lead to a decrease in oxycodone plasma concentrations, lack of efficacy or, possibly, development of an abstinence syndrome in a patient who had developed physical dependence to oxycodone. If co-administration with OxyContin Neo is necessary, monitor for signs of opioid withdrawal and consider dose adjustments until stable drug effects are achieved. After stopping the treatment of a CYP3A4 inducer, as the effects of the inducer decline, the oxycodone plasma concentration will increase which could increase or prolong both the therapeutic and adverse effects, and may cause serious respiratory depression (see Pharmacology under Actions). Mixed Agonist/Antagonist Opioid Analgesics: Mixed agonist/antagonist analgesics (i.e., pentazocine, nalbuphine, and butorphanol) and partial agonist (buprenorphine) analgesics may reduce the analgesic effect of oxycodone or precipitate withdrawal symptoms. Avoid the use of mixed agonist/antagonist and partial agonist analgesics in patients receiving OxyContin Neo. Diuretics: Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. Opioids may also lead to acute retention of urine by causing spasm of the sphincter of the bladder, particularly in men with enlarged prostates. Anticholinergics: Anticholinergics or other medications with anticholinergic activity when used concurrently with opioid analgesics may result in increased risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. Monitor patients for signs of urinary retention or reduced gastric motility when OxyContin Neo is used concurrently with anticholinergic drugs. Antihypertensive Agents: Hypotensive effects of these medications may be potentiated when used concurrently with oxycodone, leading to increased risk of orthostatic hypotension. Coumarin Derivatives: Although there is little substantiating evidence, opiate agonists have been reported to potentiate the anticoagulant activity of coumarin derivatives. Metoclopramide: Concurrent use with oxycodone may antagonise the effects of metoclopramide on gastrointestinal motility. Monoamine Oxidase Inhibitors (MAOIs): Non-selective MAOIs intensify the effects of opioid drugs which can cause anxiety, confusion and significant respiratory depression. Severe and sometimes fatal reactions have occurred in patients concurrently administered MAOIs and pethidine. Oxycodone should not be given to patients taking non-selective MAOIs or within 14 days of stopping such treatment. As it is unknown whether there is an interaction between selective MAOIs (e.g. selegiline) and oxycodone, caution is advised with this drug combination. Opioid Agonist Analgesics (including morphine, pethidine): Additive CNS depressant, respiratory depressant and hypotensive effects may occur if two or more opioid agonist analgesics are used concurrently. Caution For Usage Instructions on How Supplied/Storage and Handling: OxyContin Neo (oxycodone hydrochloride controlled-release) Tablets 10 mg are round, white-colored, bi-convex tablets debossed with OP on one side and 10 on the other and are supplied as child-resistant closure, opaque plastic bottles of 30 and 12, and unit dose packaging with 10 individually numbered tablets per card; three cards per glue end carton, two cards per glue end carton and one card per glue end carton. OxyContin Neo (oxycodone hydrochloride controlled-release) Tablets 20 mg are round, pink-colored, bi-convex tablets debossed with OP on one side and 20 on the other and are supplied as child-resistant closure, opaque plastic bottles of 30 and 12, and unit dose packaging with 10 individually numbered tablets per card; three cards per glue end carton, two cards per glue end carton and one card per glue end carton. OxyContin Neo (oxycodone hydrochloride controlled-release) Tablets 40 mg are round, yellow-colored, bi-convex tablets debossed with OP on one side and 40 on the other and are supplied as child-resistant closure, opaque plastic bottles of 30 and 12, and unit dose packaging with 10 individually numbered tablets per card; three cards per glue end carton, two cards per glue end carton and one card per glue end carton. OxyContin Neo (oxycodone hydrochloride controlled-release) Tablets 60 mg are round, red-colored, bi-convex tablets debossed with OP on one side and 60 on the other and are supplied as child-resistant closure, opaque plastic bottles of 30 and 12, and unit dose packaging with 10 individually numbered tablets per card; three cards per glue end carton, two cards per glue end carton and one card per glue end carton. OxyContin Neo (oxycodone hydrochloride controlled-release) Tablets 80 mg are round, green-colored, bi-convex tablets debossed with OP on one side and 80 on the other and are supplied as child-resistant closure, opaque plastic bottles of 30 and 12, and unit dose packaging with 10 individually numbered tablets per card; three cards per glue end carton, two cards per glue end carton and one card per glue end carton. Dispense in tight, light-resistant, child-resistant closure, opaque plastic bottles or dispense unit dose packaging with 10 individually numbered tablets per card; three cards per glue end carton, two cards per glue end carton and one card per glue end carton.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
